Project period: 2021-2025
Grant DK kr. 3.39 mio
A key element in the green transition to a fossil free society is that we can utilize electricity efficiently. Everything cannot be electrified, and we therefore need to store energy in the form of chemical energy in different fuels and chemicals. An essential step of electrifying the production of fuels and chemicals (Power-to-X) is water splitting to produce hydrogen. Reacting this hydrogen with CO or CO2 enables us to produce synthetic fuels or chemicals with the potential to replace fossil-based resources.
The most common method of splitting water today is through electrolysis, but this process is still associated with considerable energy losses. Thermal two-step water splitting may offer an alternative route towards upscaling and increasing the efficiency of water splitting. The principle for two-step water spitting is that a metal oxide is heated to high temperature where the oxygen desorbs and the material is reduced. Subsequently the material is cooled, and by adding water the material will be oxidized again, the hydrogen is evolved and the material will be back at its original state. This cycle is then repeated.
This will be done by linking our knowledge of catalyst materials and surface science with that of electromagnetic materials to develop a new cyclic process where the energy for water splitting is supplied by heat, i.e., electrified thermal water splitting. The principle is illustrated below.
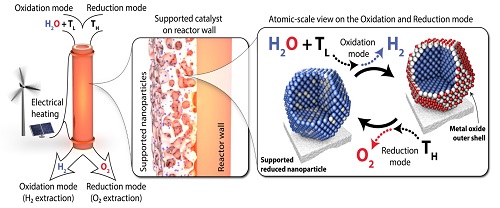
The scope of this project is to develop:
- A new process where the reaction energy is provided by electric heating (either resistive or inductive), enabling energy to be dissipated quickly and directly within the materials, and production to be flexible using surplus electricity.
- New materials where surface processes are used to split the water thermally rather than bulk. This enables the fast heating technology. The principles could also work for CO2 splitting (carbon capture), and in the longer run the two processes could be combined for fuel production.